Modeling Electron Cloud 4.18.12
Lithium Atom
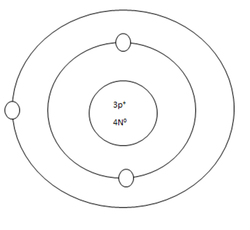
Protons=3
Neutrons=4
Electrons=3
Positive Charges in atom=3
Negative Charges in atom=3
Energy level filled in atom=2
Is energy level full=no
Neutrons=4
Electrons=3
Positive Charges in atom=3
Negative Charges in atom=3
Energy level filled in atom=2
Is energy level full=no
Beryllium Atom
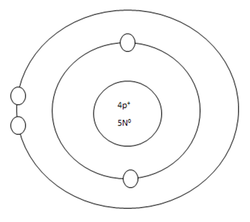
Protons=4
Neutrons=5
Electrons=4
Positive Charges in atom=4
Negative Charges in atom=4
Energy level filled in atom=2
Is energy level full=no
Neutrons=5
Electrons=4
Positive Charges in atom=4
Negative Charges in atom=4
Energy level filled in atom=2
Is energy level full=no
Carbon Atom
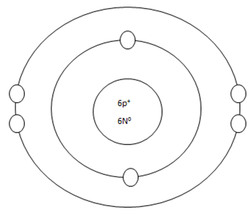
Protons=6
Neutrons=6
Electrons=6
Positive Charges in atom=6
Negative Charges in atom=6
Energy level filled in atom=2
Is energy level full=no
Neutrons=6
Electrons=6
Positive Charges in atom=6
Negative Charges in atom=6
Energy level filled in atom=2
Is energy level full=no
Oxygen Atom
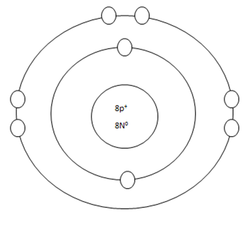
Protons=8
Neutrons=8
Electrons=8
Positive Charges in atom=8
Negative Charges in atom=8
Energy level filled in atom=2
Is energy level full=no
Neutrons=8
Electrons=8
Positive Charges in atom=8
Negative Charges in atom=8
Energy level filled in atom=2
Is energy level full=no
Neon Atom
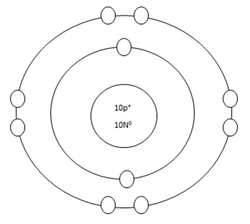
Protons=10
Neutrons=10
Electrons=10
Positive Charges in atom=10
Negative Charges in atom=10
Energy level filled in atom=2
Is energy level full=yes
Neutrons=10
Electrons=10
Positive Charges in atom=10
Negative Charges in atom=10
Energy level filled in atom=2
Is energy level full=yes
Sodium Atom
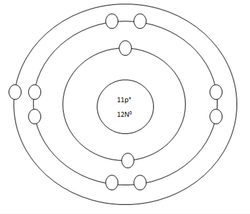
Protons=11
Neutrons=12
Electrons=11
Positive Charges in atom=11
Negative Charges in atom=11
Energy level filled in atom=3
Is energy level full=no
Neutrons=12
Electrons=11
Positive Charges in atom=11
Negative Charges in atom=11
Energy level filled in atom=3
Is energy level full=no
Aluminum Atom
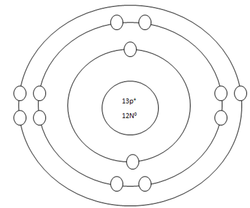
Protons=13
Neutrons=14
Electrons=13
Positive Charges in atom=13
Negative Charges in atom=13
Energy level filled in atom=3
Is energy level full=no
Neutrons=14
Electrons=13
Positive Charges in atom=13
Negative Charges in atom=13
Energy level filled in atom=3
Is energy level full=no
Phosphorus Atom
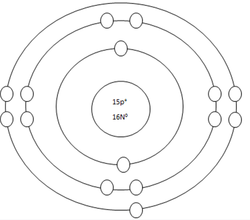
Protons=15
Neutrons=16
Electrons=15
Positive Charges in atom=15
Negative Charges in atom=15
Energy level filled in atom=3
Is energy level full=no
Neutrons=16
Electrons=15
Positive Charges in atom=15
Negative Charges in atom=15
Energy level filled in atom=3
Is energy level full=no
Sulfur Atom
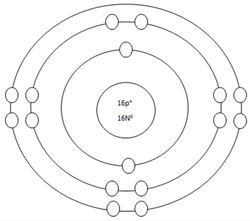
Protons=16
Neutrons=16
Electrons=16
Positive Charges in atom=16
Negative Charges in atom=16
Energy level filled in atom=3
Is energy level full=no
Neutrons=16
Electrons=16
Positive Charges in atom=16
Negative Charges in atom=16
Energy level filled in atom=3
Is energy level full=no
Chlorine Atom
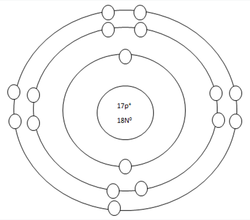
Protons=17
Neutrons=18
Electrons=17
Positive Charges in atom=17
Negative Charges in atom=17
Energy level filled in atom=3
Is energy level full=no
Neutrons=18
Electrons=17
Positive Charges in atom=17
Negative Charges in atom=17
Energy level filled in atom=3
Is energy level full=no
Argon Atom
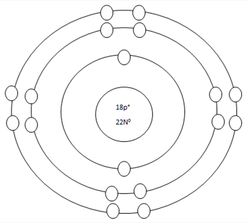
Protons=18
Neutrons=18
Electrons=18
Positive Charges in atom=18
Negative Charges in atom=18
Energy level filled in atom=3
Is energy level full=yes
Neutrons=18
Electrons=18
Positive Charges in atom=18
Negative Charges in atom=18
Energy level filled in atom=3
Is energy level full=yes
Calcium Atom
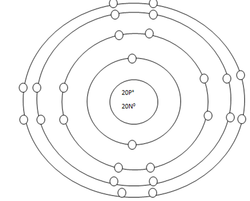
Protons=20
Neutrons=20
Electrons=20
Positive Charges in atom=20
Negative Charges in atom=20
Energy level filled in atom=4
Is energy level full=no
Neutrons=20
Electrons=20
Positive Charges in atom=20
Negative Charges in atom=20
Energy level filled in atom=4
Is energy level full=no
1. How many valance electrons are found in a neutral atom of beryllium?
There are 2 in Beryllium.
2. How many valance electrons are found in a neutral atom of nitrogen?
There are 5 in Nitrogen.
3. How many valance electrons are found in a neutral atom of oxygen?
There are 6 in Oxygen.
4. How many valance electrons are found in a neutral atom of neon?
There are 8 in Neon.
5. Is the valance shell of neon full?
Yes it is.
6. How many valance electrons are found in a neutral atom of sodium?
There are 1 in Sodium.
7. How many valance electrons are found in a neutral atom of magnesium?
There are 2 in Magnesium.
8. How many valance electrons are found in a neutral atom of chlorine?
There are 7 in Chlorine.
9. How many valance electrons are found in a neutral atom of argon?
There are 7 in Chlorine.
10. Is the valance shell of argon full?
Yes, it is full.
There are 2 in Beryllium.
2. How many valance electrons are found in a neutral atom of nitrogen?
There are 5 in Nitrogen.
3. How many valance electrons are found in a neutral atom of oxygen?
There are 6 in Oxygen.
4. How many valance electrons are found in a neutral atom of neon?
There are 8 in Neon.
5. Is the valance shell of neon full?
Yes it is.
6. How many valance electrons are found in a neutral atom of sodium?
There are 1 in Sodium.
7. How many valance electrons are found in a neutral atom of magnesium?
There are 2 in Magnesium.
8. How many valance electrons are found in a neutral atom of chlorine?
There are 7 in Chlorine.
9. How many valance electrons are found in a neutral atom of argon?
There are 7 in Chlorine.
10. Is the valance shell of argon full?
Yes, it is full.
