Reactivity 4.20.12
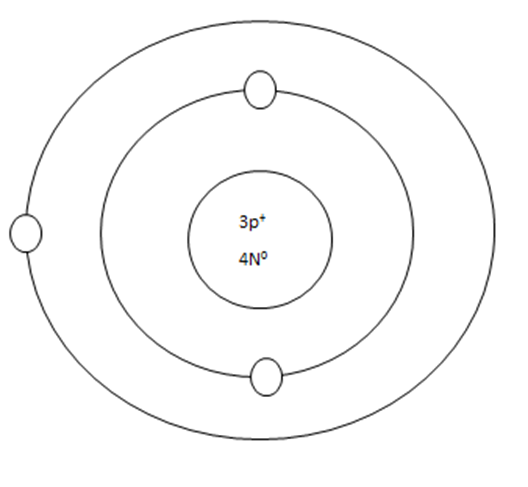
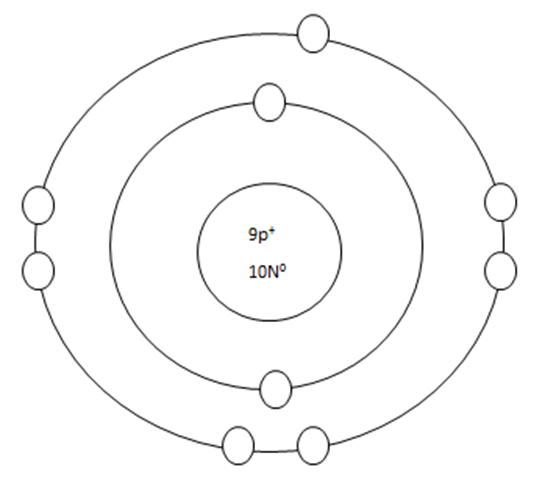
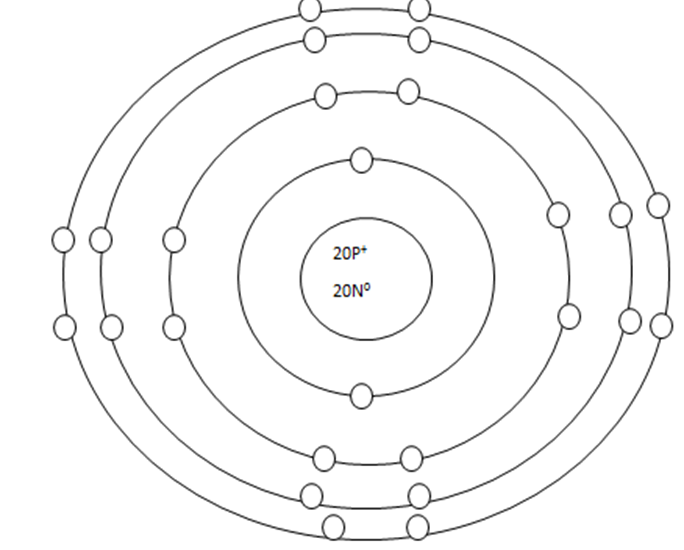
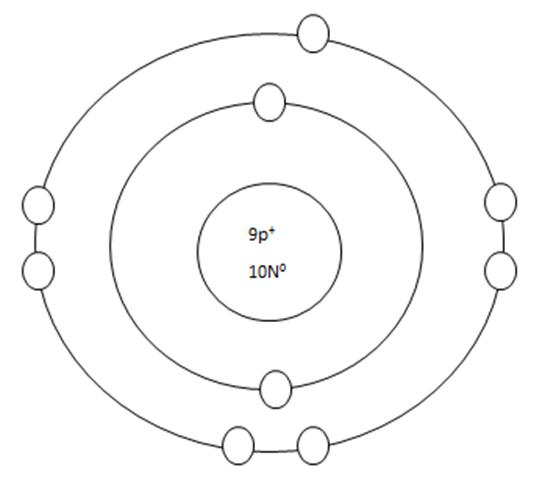
As it turns out, the key number for the valance shell is the number "8". The number "2" is also a key number but only for the atoms hydrogen, helium, lithium, beryllium, and boron. So, what makes the 8, and sometimes 2, key numbers?
Scientists learned a simple rule of thumb about atoms. They tend to combine to that their valance shells are full of electrons. This is called the octet rule. (Not the root oct = 8 in the word octet, like octopus!)
This means two things:
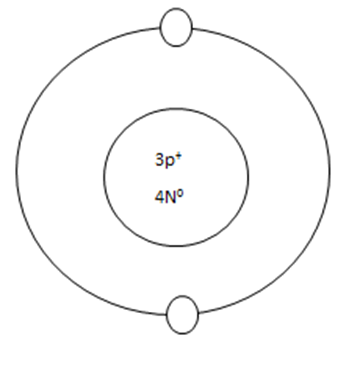
1: If a neutral atom's valance shell is full, then the atom will not react with other atoms. (Noble gasses in group 18)
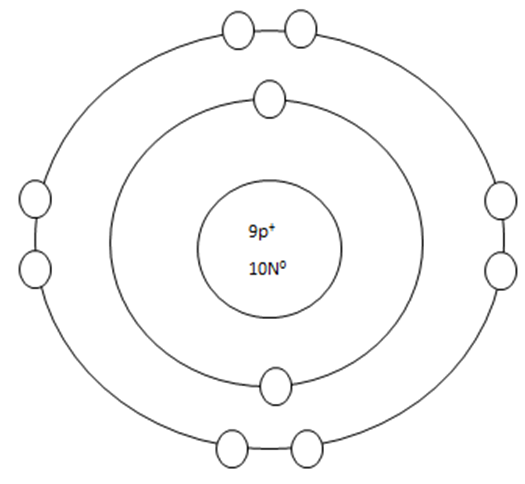
2. If a neutral atom's valance shell has space available, then the atom will react with other atoms based on the number of electrons found in the valance shell.
Some atoms only need 1, 2, or 3 more electrons to fill the valance shell. These electrons come from other atoms when they react.
Some atoms need to remove 1, 2, or 3 electrons to have the energy layer below become the valance shell that is already full. The removes electrons are accepted by other atoms when they react.
Some atoms tend to react with other atoms in a way electrons are shared rather than giving up or receiving electrons.
WHEN AN ATOM HAS A CHARGE, IT BECOMES AN ION.
WHEN ATOMS REACT, THEY EITHER GIVE UP A VALANCE ELECTRON, GAIN A VALANCE ELECTRON, OR SHARE A VALANCE ELECTRON.Groups that LOSE: 1, 2, 3 GAIN: 5, 6, 7, 15, 16, 17 SHARE 14.
Scientists learned a simple rule of thumb about atoms. They tend to combine to that their valance shells are full of electrons. This is called the octet rule. (Not the root oct = 8 in the word octet, like octopus!)
This means two things:
1: If a neutral atom's valance shell is full, then the atom will not react with other atoms. (Noble gasses in group 18)
2. If a neutral atom's valance shell has space available, then the atom will react with other atoms based on the number of electrons found in the valance shell.
Some atoms only need 1, 2, or 3 more electrons to fill the valance shell. These electrons come from other atoms when they react.
Some atoms need to remove 1, 2, or 3 electrons to have the energy layer below become the valance shell that is already full. The removes electrons are accepted by other atoms when they react.
Some atoms tend to react with other atoms in a way electrons are shared rather than giving up or receiving electrons.
WHEN AN ATOM HAS A CHARGE, IT BECOMES AN ION.
WHEN ATOMS REACT, THEY EITHER GIVE UP A VALANCE ELECTRON, GAIN A VALANCE ELECTRON, OR SHARE A VALANCE ELECTRON.Groups that LOSE: 1, 2, 3 GAIN: 5, 6, 7, 15, 16, 17 SHARE 14.