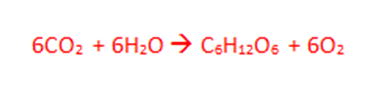Warm Ups 4.12.12
4.12.12
What is matter?
Matter is anything that has mass and takes up space in one of these three states: solid, liquid, or gas.
4.16.12
Where is the mass of the atom found? What is the mass of this atom?
In the nucleus. The mass is 6 amu. (atomic mass unit).
4.17.12
What is the charge of the atom?
Neutral.
What is the mass of the atom?
13 amu.
What is it's atomic number?
6. (6 protons).
What element is the atom?
Carbon.
4.18.12
How many protons are in this sodium atom?
11 protons.
How many electrons are in this atom?
11 electrons
How many neutrons are in this sodium atom?
12 neutrons.
Draw a diagram of this atom.
4.19.12
Draw a diagram of a chlorine atom.
Draw a diagram of a chlorine atom.
4.20.12
Draw the atom of potassium. How many valence electrons does this atom have?
1 valance electron.
Draw the atom of potassium. How many valence electrons does this atom have?
1 valance electron.
4.23.12
Draw a magnesium atom. How many valence electrons does this atom have? Is it stable, if not, what will happen to its charge?
4.24.12
The magnesium atom has 2 valence electrons, it is not closed, so it is not stable. It has a positive charge, because it will lose the extra negative electrons. Positive 2 charge.
4.25.12
1. How many elements are in the reaction above? (This shows photosynthesis.)
3 elements: Carbon, Oxygen, and Hydrogen.
2. How many atoms are in the products of the reaction?
36 Atoms.
3. Does this equation violate the law of conservation of matter?
No, the masses are both the same.
5.1.12
Groups or families on the periodic table have what in common? (COLUMNS)
They each have the same number of valence electrons.
Periods on the periodic table have what in common? (ROWS)
They are in line with their energy levels.
5.2.12
What is the atomic number of the element above?
3.
What is the atomic mass of the element above?
7.
Is the element an ion? Explain.
No, because is has 3 protons and 3 electrons.
5.3.12
In my neutral state, I have 35 electrons. What element am I?
Bromine.
When I have a charge of +2 , I have 38 electrons. What element am I?
Zirconium.
5.7.12
How many elements and atoms are in each molecule?
Hg3(PO4)2
There are 3 elements, Hg, Phosphorous, and Oxygen. There are 13 atoms.
3PO4
There are 2 elements, Phosphorous and Oxygen. There are 15 atoms.
Na3AsO3
3 elements, Na, As, and O. There are 7 atoms.
5.8.12
Balance this equation. HIO3 + FeI2 +HCl --> FeCl3 + ICl + H2O
5HIO3 + 4FeI2 + 25HCl --> 4FeCl3 + 13ICl + 15H2O
5.10.12
Balance this equation: _H2SO4 + _Pb(OH)4 --> _Pb(SO4)2 + _H2O
2H2SO4 + Pb(OH)4 --> Pb(SO4)2 + 4H2O
C12H22O11 + KClO3 --> CO2 + H2O + KCl
C12H22O11 + 8KClO3 --> 12CO2 + 11H2O + 8KCl
5.11.12
Balance this equation. _Ca3(PO4)2 + _ H2SO4 --> _CaSO4 + _Ca(H2PO4)2
Ca3(PO4)2 + 2H2SO4 = 2CaSO4 + Ca(H2PO4)2
5.15.12
Balance this equation. _Fe + _H2SO4 --> _Fe2(SO4)3 + _H2
2Fe + 3H2SO4 --> 1Fe2(SO4)3 + 3H2
1. How many elements are in the reaction above? (This shows photosynthesis.)
3 elements: Carbon, Oxygen, and Hydrogen.
2. How many atoms are in the products of the reaction?
36 Atoms.
3. Does this equation violate the law of conservation of matter?
No, the masses are both the same.
5.1.12
Groups or families on the periodic table have what in common? (COLUMNS)
They each have the same number of valence electrons.
Periods on the periodic table have what in common? (ROWS)
They are in line with their energy levels.
5.2.12
What is the atomic number of the element above?
3.
What is the atomic mass of the element above?
7.
Is the element an ion? Explain.
No, because is has 3 protons and 3 electrons.
5.3.12
In my neutral state, I have 35 electrons. What element am I?
Bromine.
When I have a charge of +2 , I have 38 electrons. What element am I?
Zirconium.
5.7.12
How many elements and atoms are in each molecule?
Hg3(PO4)2
There are 3 elements, Hg, Phosphorous, and Oxygen. There are 13 atoms.
3PO4
There are 2 elements, Phosphorous and Oxygen. There are 15 atoms.
Na3AsO3
3 elements, Na, As, and O. There are 7 atoms.
5.8.12
Balance this equation. HIO3 + FeI2 +HCl --> FeCl3 + ICl + H2O
5HIO3 + 4FeI2 + 25HCl --> 4FeCl3 + 13ICl + 15H2O
5.10.12
Balance this equation: _H2SO4 + _Pb(OH)4 --> _Pb(SO4)2 + _H2O
2H2SO4 + Pb(OH)4 --> Pb(SO4)2 + 4H2O
C12H22O11 + KClO3 --> CO2 + H2O + KCl
C12H22O11 + 8KClO3 --> 12CO2 + 11H2O + 8KCl
5.11.12
Balance this equation. _Ca3(PO4)2 + _ H2SO4 --> _CaSO4 + _Ca(H2PO4)2
Ca3(PO4)2 + 2H2SO4 = 2CaSO4 + Ca(H2PO4)2
5.15.12
Balance this equation. _Fe + _H2SO4 --> _Fe2(SO4)3 + _H2
2Fe + 3H2SO4 --> 1Fe2(SO4)3 + 3H2