Periodic Table and Chemical Reaction Notes 4.24.12
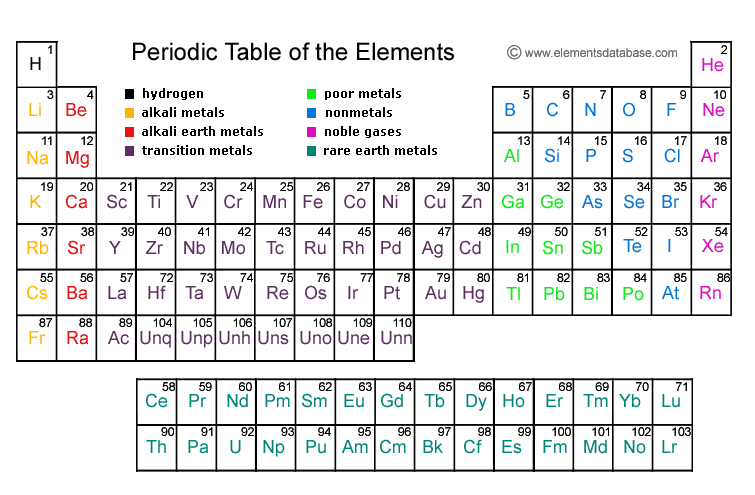
7 rows
-called periods
-each period represents the number of occupied energy levels in the row
18 columns
-called groups or families
-share similar chemical and physical properties
-have the same amount of valence electrons
http://www.elementsdatabase.com/
-If it touches the zigzag line it is a metalliod.
-To the right od the zigzag lines are non metals.
-To the left of the zigzag line are metals.
-called periods
-each period represents the number of occupied energy levels in the row
18 columns
-called groups or families
-share similar chemical and physical properties
-have the same amount of valence electrons
http://www.elementsdatabase.com/
-If it touches the zigzag line it is a metalliod.
-To the right od the zigzag lines are non metals.
-To the left of the zigzag line are metals.
|
|
Chemical Reaction Notes:
-you know a chemical reaction has happened when a product is made
-matter can't be created or destroyed
-Example: C2H4O2 + NaHCO3 Reaction: H2O + NaO2C2H3 + CO2
-you know a chemical reaction has happened when a product is made
-matter can't be created or destroyed
-Example: C2H4O2 + NaHCO3 Reaction: H2O + NaO2C2H3 + CO2